
"Using technology to design the most efficient next-generation breast implants on the market on all the quality / sucess criteria expeted by surgeons and patients"

PRYME® Technology featuring Estyme® Matrix
New design methods trough Finite Elements Modeling (FEM) to predict the implant behavior

ESTYME® MATRIX ROUND Smooth
Smooth Silicone Gel-Filled Breast Implant with large references choice adapted for demanding surgeons and patients
ESTYME® MATRIX concept for the whole breast implants portfolio
A matrix range to fullfil patients specific needs in aesthetics and reconstructive surgery

Pre market clinical trials

1
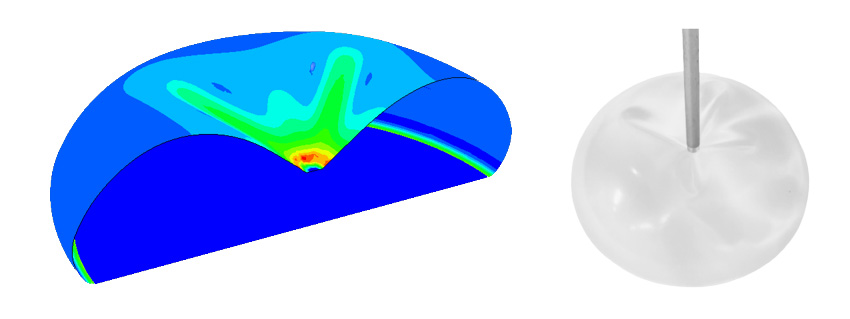
- A digital implant using finite element calculations
- Simulation of extreme stresses on our implants to optimise their design.
- Our concern is to improve the performance and safety of our breast implants in vitro.
2
- A proprietary know how in the silicone transformation process to get the best of this perfectly biocompatible and FDA-certified material
- Unique machines imagined and designed by SYMATESE AESTHETICS
- Extensive expertise in breast implants.
Combined properties regarding the shell and gel
Help to improve the longevity of our implants 5,6 and also facilitate their insertion and placement. 7,8,9
Viscoelasticity, cohesiveness and cross-linking properties, resulting from a unique know-how.
The results of tests of resistance10,11,12 and diffusion of the gel through the shell «also called Bleed» 13,14,15,16,17 demonstrate the excellent level of performance of our products. By reducing this diffusion, we limit the risk of inflammation and consequently the risk of capsular contracture.18

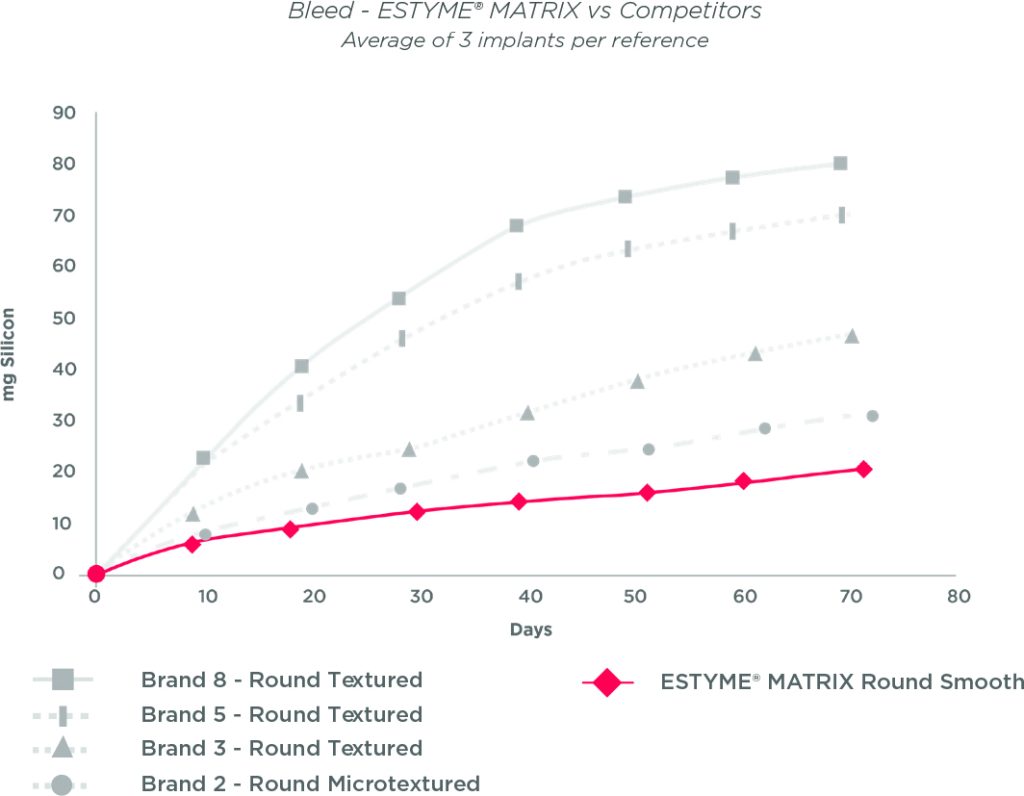
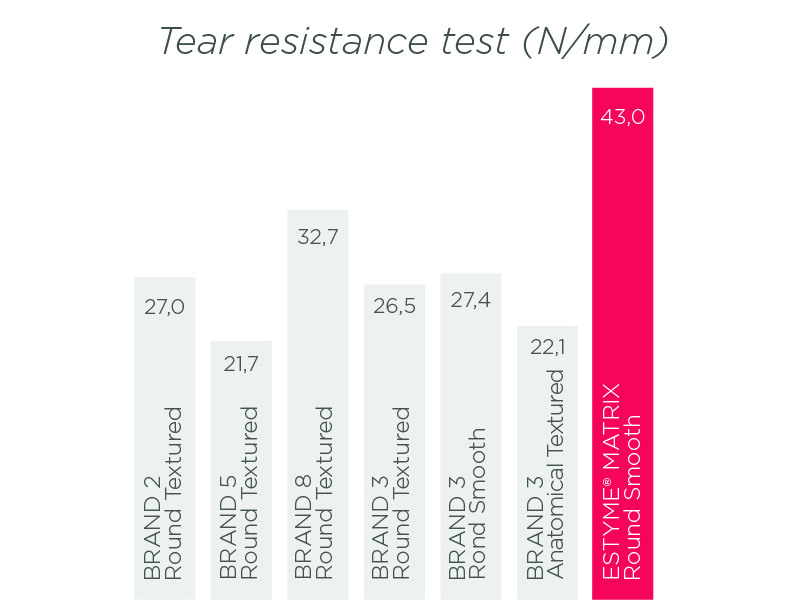
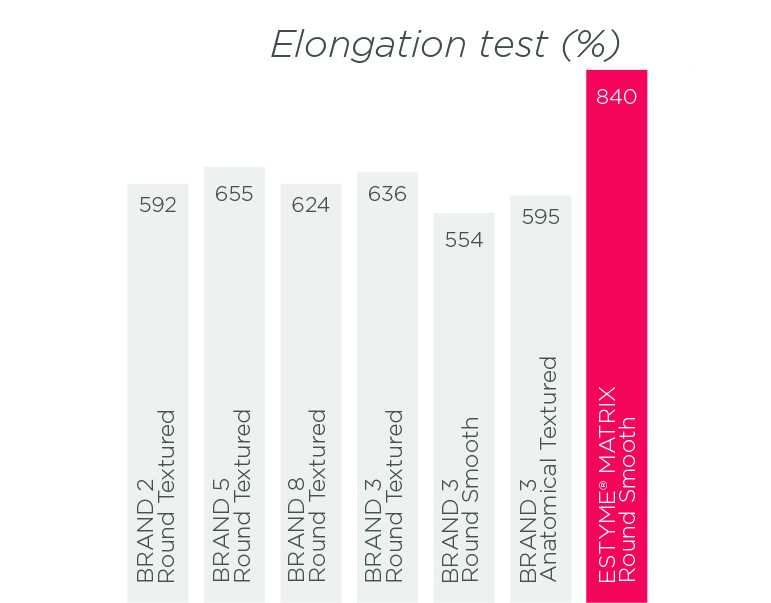

ESTYME® MATRIX Round Smooth
The Breast implants are used in a surgical procedure :
- To replace breast tissue that has been removed because of cancer or injury (breast reconstruction), or to replace breast tissue that has failed to develop properly because of a severe breast abnormality.
- To increase the size of the breast(s) (breast augmentation).
Potential benefits to patients implanted with the device include the following :
- Better quality of life.
- Increase of self esteem and self image.
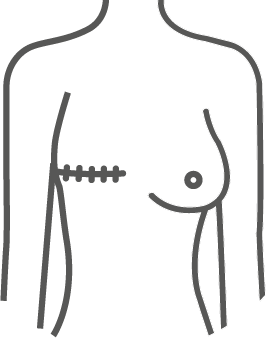
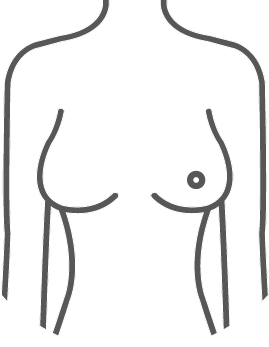
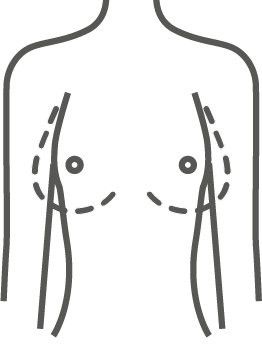
ESTYME® MATRIX concept
The wide variety of sizes and volumes allows the surgeon to respond specifically to the expectations and needs of the patient for natural and «customized» results.
- Optimized algorithms and increments
- Linear projection according to the base
- More than 100 references


For one base, 5 profiles, up to 21 Diameters for each :
Prospective EMMIE study
This completed study has evaluated the safety and performance of the ESTYME® MATRIX Round and Anatomical Silicone Gel-Filled Breast Implants in the breast augmentation and reconstruction at 3 months, 1 year and 2 years post-procedure.
Performance evaluation by the surgeon at 2 years:
- 100% SATISFACTION.
- Success of the implantation procedure.
- Overall satisfaction, especially with size, shape, firmness and appearance.
Performance evaluation by the patient at 2 years :
- Overall satisfaction: 100%
- Quality of life evaluation: Improved appearance and self-esteem
Also an on-going study EMMA Study :
- Evaluation of the Safety and Performance of ESTYME© MATRIX
- Round microtextured Breast Implants in augmentation
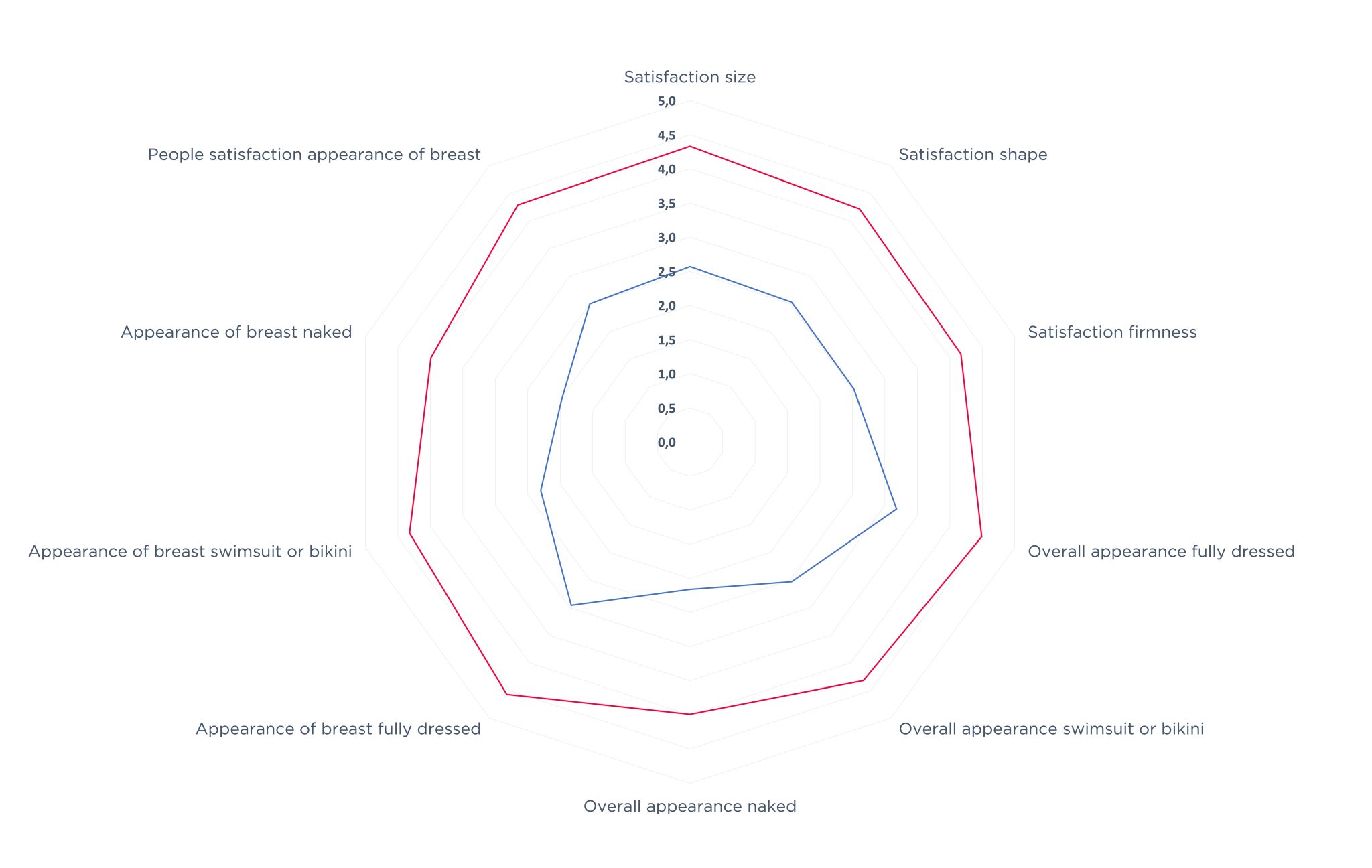
Patient evaluation of satisfaction
The satisfaction in terms of shape, consistency of the breast and quality of life is increased at 2 years compared to before surgery improving his silhouette and self esteem.
- Before surgery
- 2 years
5 Maxwell, 2017 : Breast implant design
6 Hillard, 2017: Silicone breast implant rupture: a review
7 Internal report, RR&D17003 Round Smooth
8 Clinical investigation report: CLIN PMI-002 EMMIE STUDY
9 internal report PMI19 031 V0: Summary for insertion result of Estyme® Matrix silicone gel-filled breast implants during EMMIE clinical study.
10 Internal report, RR&D17003 Round smooth
11 LNE report P158262 for Eurosilicone, Sebbin, Establishment lab , Allergan
12 LNE reports P142278, P150437 and P157512 for Mentor Round textured, round smooth and anatomical
14 LNE report n°P169625 dated from July 2016 (Estyme matrix round smooth)
16 LNE report n°P149805 dated from (competitors allergan Motiva Sebbin Eurosilicone)
17 LNE report n°P142278 dated from (competitor Mentor)

