
Unique Syringe-Gel combination

Efficacy and safety proven with clinical trials

C-HA.PURE TECHNOLOGY™ by SYMATESE

ESTYME® LIFT
SYMATESE HA KNOW-HOW recognised by leaders in various fields
15 years scientific research in HA applied in facial rejuvenation and therapeutic areas
Lipoatrophy HIV Radiotherapy Ophthalmology Rheumatology
New augmented HA technology
Our milestones in medical aesthetics
- 2011 : 1st generation HA dermal Fillers launched by a world leader
- 2021 : 2nd generation : SYMATESE gains the CE marking for ESTYME LIFT® among its future 4 products range
- 2022 : ANVISA authorization for ESTYME® LIFT Medical device Class IV (ANVISA N°10245850013).
C-HA.PURE TECHNOLOGY™ by SYMATESE is an exclusive COLD temperature cross-linking process which is gentler and requires less BDDE. More benefits to be revealed soon.
HIGH TEMPERATURE
TYPICAL CROSSLINKING
COLD TEMPERATURE
C-HA.PURE TECHNOLOGYTM by SYMATESE
ESTYME® LIFT presentation
Our cold technology HA allowed to develop targeted products, each dedicated to an indication of the face.
Each product of ESTYME® FILLERS range, including ESTYME® LIFT has specific characteristics and responds exactly and effectively to the needs of the area concerned.
ESTYME® LIFT¹ is a sterile, biodegradable, transparent gel made of cross-linked hyaluronic acid from non-animal sources containing lidocaine hydrochloride 3 mg / ml
- The gel is presented in a 1 mL plastic syringe.
- The gel in the syringe is sterilized by moist heat.
- One syringe and two ultra-thin-walled needles are packaged in a blister.
- The needles supplied are sterilized by irradiation. ESTYME® LIFT is available in a box of one unit.
- The device (syringe, gel and needles) is for single use.
For more information, read the instruction for use
¹According to ESTYME® LIFT instructions for use
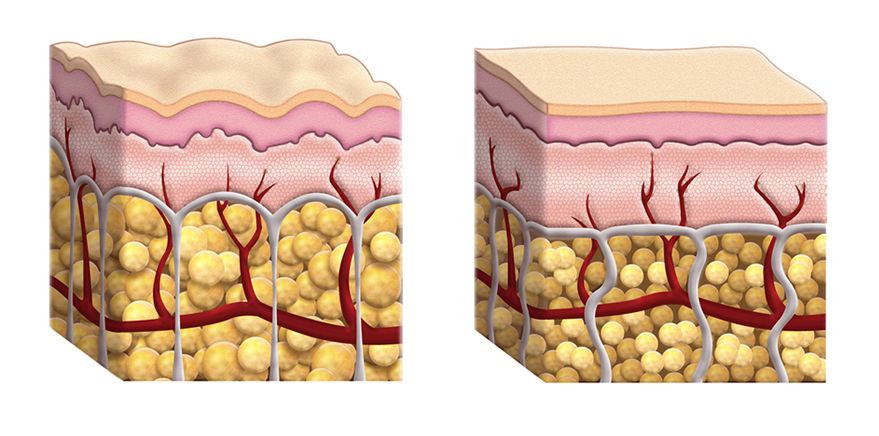
ESTYME® LIFT mode of action
HA ESTYME® FILLERS range
ESTYME® LIFT acts by adding volume to the tissue, thereby restoring the skin contours and the volume and shape of the face to the desired level. The volume and lifting capacity originate from the ability of the cross-linked hyaluronic acid gel to hold its shape in the tissue, thus maintaining over time the volume and projection obtained at injection.
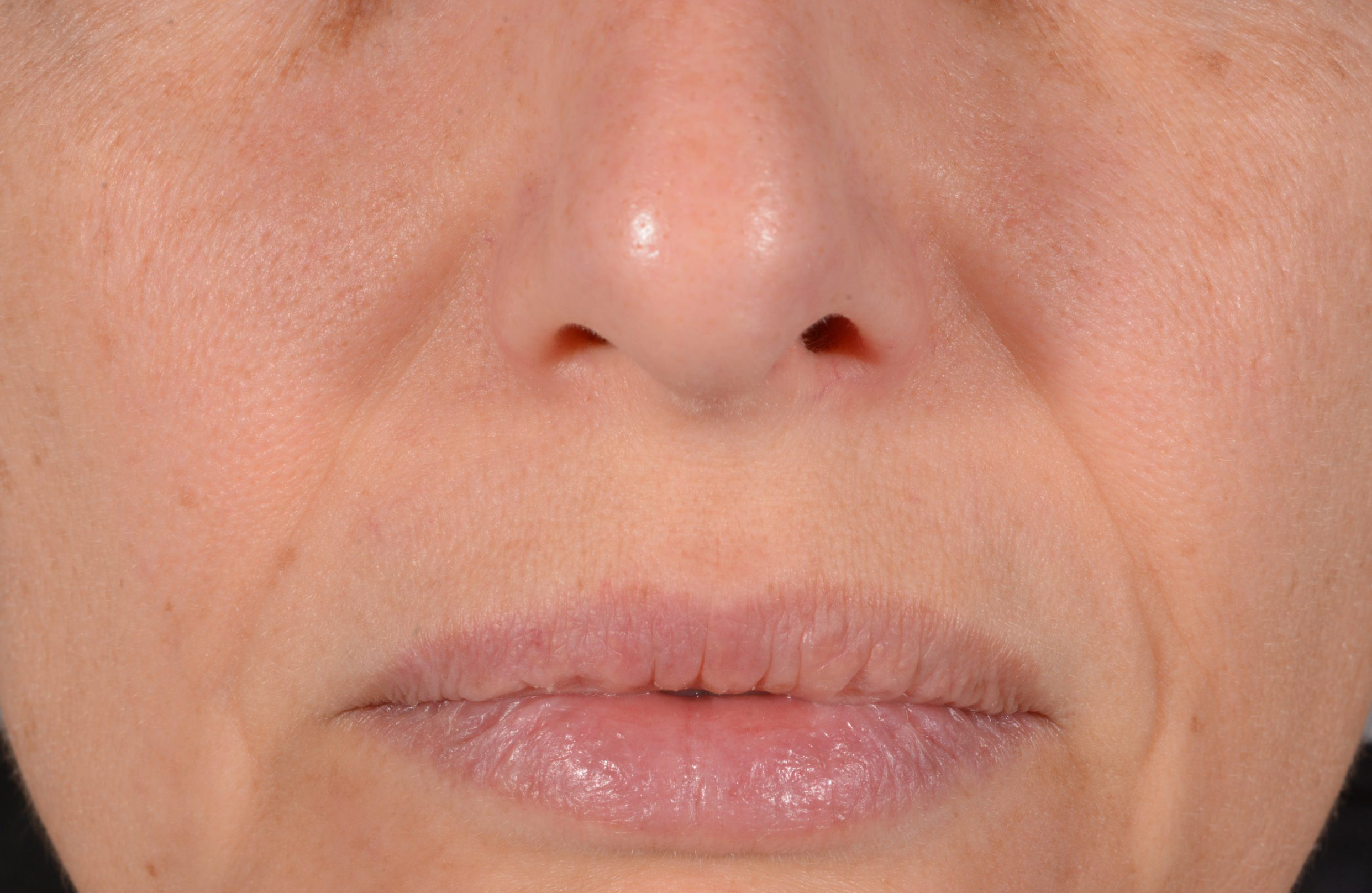
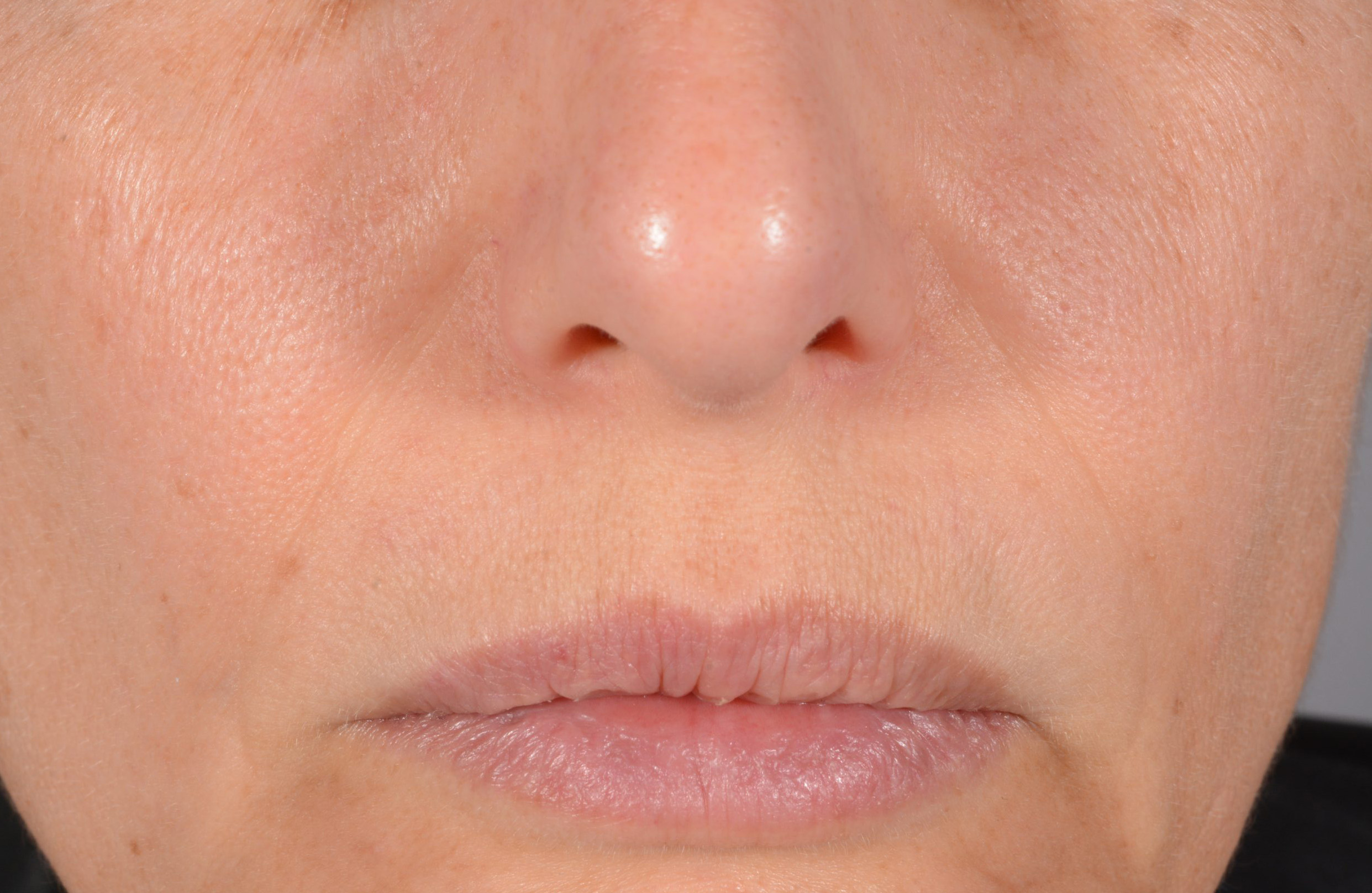
Before and after
with ESTYME® LIFT on NASIOLABIAL FOLDS
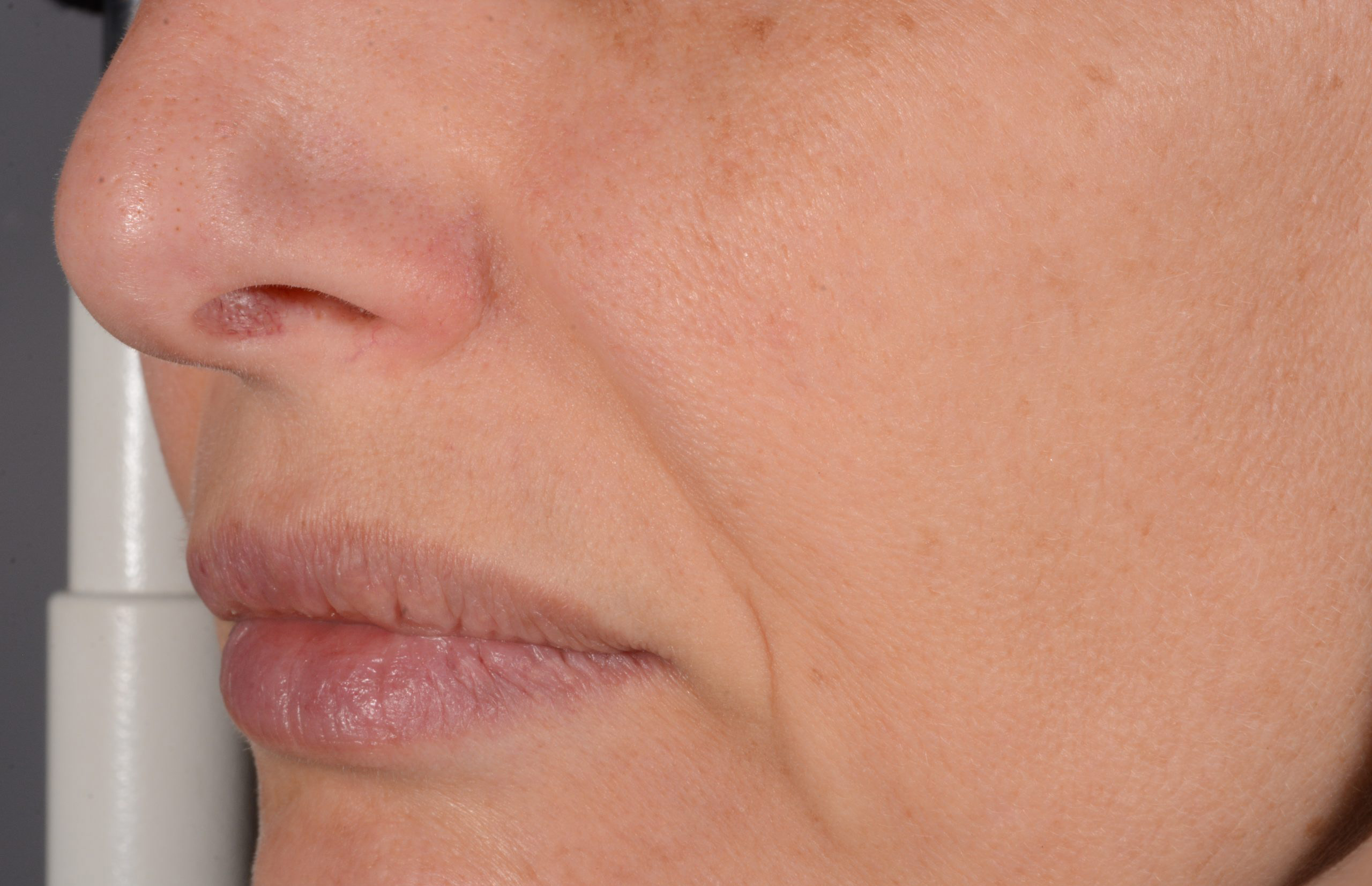
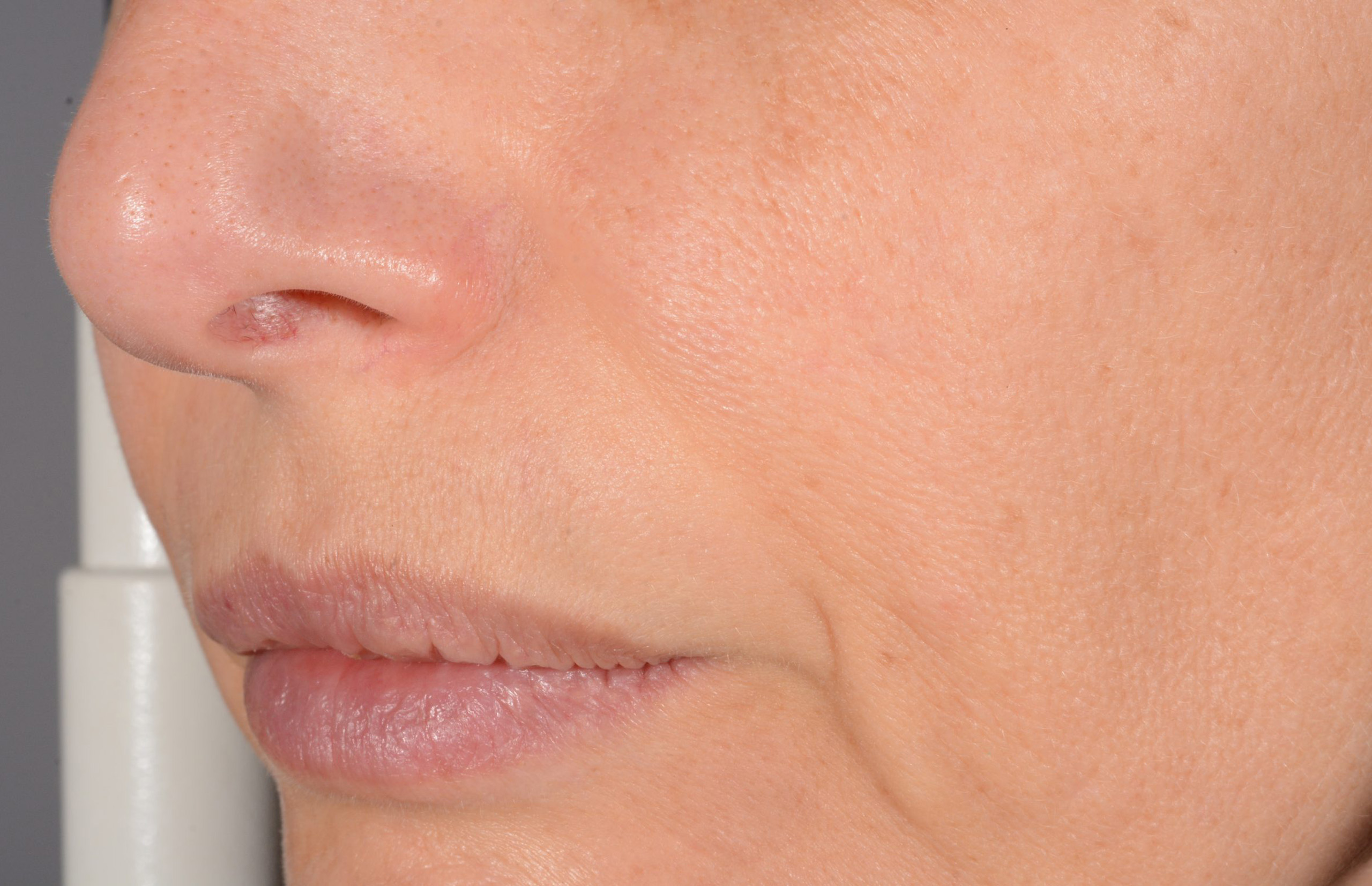

Symatese developed and manufactured both syringes and HA gel for an ergonomic experience
Thanks to the combination gel-syringe by the sister companies which allows to offer a unique HA dermal filler
Many clinical studies have been performed regarding ESTYME® LIFT
Those studies are part of a robust clinical trial program, on all ESTYME® FILLERS products, demonstrating TOLERANCE, PERFORMANCE and LONG-LASTING EFFECT with 269 patients in Europe and 854 patients enrolled in China and USA.
See below the findings of the clinical trial conducted on ESTYME® LIFT in Europe
Including one comparative randomized study on ESTYME® LIFT
- 45 patients with symmetric nasolabial folds
- from moderate (grade 3) to severe (grade 4), according to the WSRS* scale.
- The mean age of subjects was 58 years
- 84% of subjects were women.
The study compares ESTYME® LIFT to a worldwide commonly used HA-lidocaine filler, approved in treatment of nasolabial folds (control). For each patient, both products are injected either on the left or on the right side.
*WSRS : Wrinkle Severity Rating Scale.
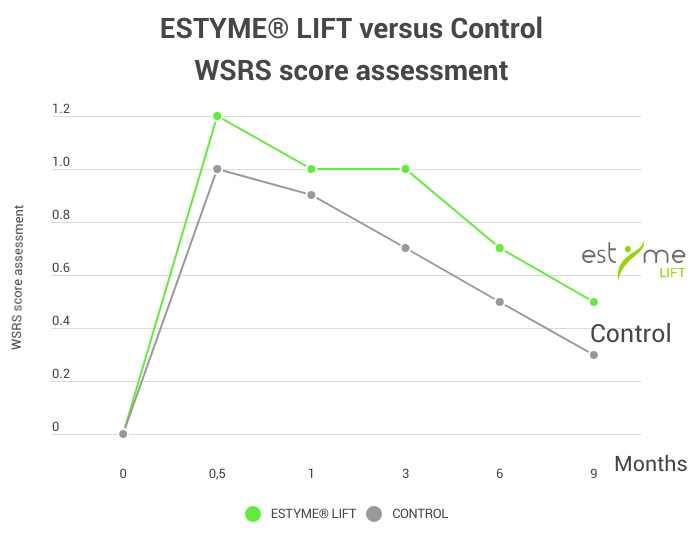
The WSRS assessment highlights an improvement in age-related folds throughout the study with a significant difference at 3 and 6 months in favor of ESTYME® LIFT as well as prolonged effectiveness by 3 months compared to the control.
Regarding safety, no significant difference was observed between both devices


